Chemistry is the study of matter, its properties, and the changes it undergoes. It is called the Central Science because there is hardly any aspect of life untouched by it. It taps into various fields of science, such as complex studies such as Pharmacology, Toxicology, and Medicine and even explains simple daily activities such as cooking and cleaning.
What is Matter?
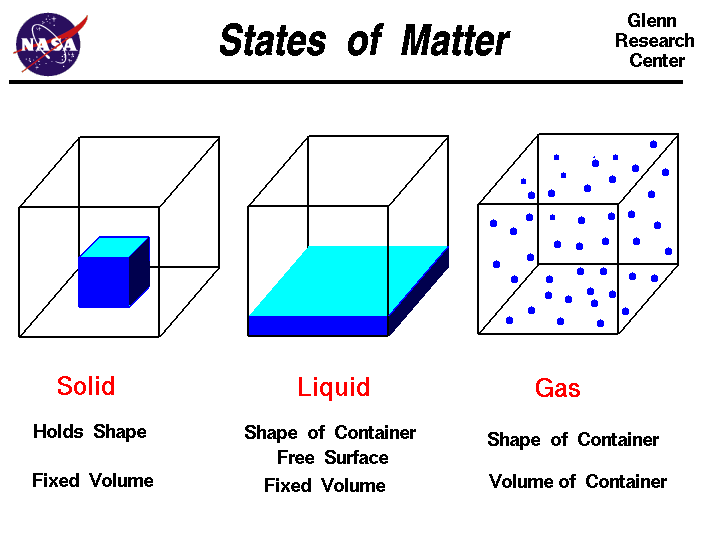 Matter is what makes up material objects; it is anything that takes up space and has mass, meaning it could be weighed. It contains atoms and molecules, which are constantly moving. The amount of energy (or movement) that atoms and molecules have influences their interaction with each other.
Matter is what makes up material objects; it is anything that takes up space and has mass, meaning it could be weighed. It contains atoms and molecules, which are constantly moving. The amount of energy (or movement) that atoms and molecules have influences their interaction with each other.The three common states of matter are solid, liquid, and gas. Solids are ordinary objects which retains its shape and volume no matter its location, due to its particles being tightly packed together and in fixed positions. Liquids occupy a definite volume but assumes the shape of the occupied portion of its container. This is so because its particles are close together, but are free to move about. Gasses matains neither its shape or volume. It expands to fill completely the container it occupies. Gases flows and are easily compressed, due to its particles moving in a rapid random motion and being far apart. The other two states of matter are plasma and Bose-Einstein Condensates. Plasmas are hot, ionized gases, formed under conditions of extremely high energy, so high, in fact, that molecules are ripped apart and only free atoms exist. Bose-Einstein Condensates is when all the atoms of the condensate attain the same quantum-mechanical state and can flow past one another without friction. These condensates are gaseous superfluids cooled to temperatures very near absolute zero.
ASPECTS OF MATTER
1. Changes
Response to a reaction; to form, to create.
2. Composition
Elements present or found in a piece of matter.
3. Application
How is is used and how it is beneficial.
4. Laws and Principles
The reason behind the change or observation.
5. Characteristics
Structure and form.
A BRIEF OUTLINE ON THE HISTORY OF CHEMISTRY
Prehistoric Chemistry
People used fire to bring about change.
2, 500 years ago
Greek philosophers were the first to formulate theories--explanations on the behavior of matter.
C. E. 500 - 1500
The experimental roots of chemistry, Alchemy (a mixture pf magic and chemistry), was propagated. Alchemists sought after the philosopher's stone, that would turn cheapers metals into gold, and an elixir that would confer immortality to those exposed to it.
The experimental roots of chemistry, Alchemy (a mixture pf magic and chemistry), was propagated. Alchemists sought after the philosopher's stone, that would turn cheapers metals into gold, and an elixir that would confer immortality to those exposed to it.
Middle of 20th Century
Francis Bacon, a judge, had the Baconian Dream, where in science could solve the world's problems.
Modern Chemistry
Chemists can now transform matter in ways that would astound alchemists, like changing crude oil to plastics, fibers, pesticide, drugs, and detergents.
FIVE MAJOR BRANCHES IN CHEMISTRY
 1. Organic Chemistry
1. Organic Chemistry Organic chemistry deals with all carbon compounds. Carbon is the basis of all life. This is important to petrochemica, pharmaceutical, and textile industries. This also studies living organisms, for they are made of cells.
2. Inorganic Chemistry
Inorganic chemistry deals with non-carbon compounds. Most acids, bases and salts are non-carbon based. Mineral compounds are also included in inorganic chemistry. Hydrochloric acid, sodium metal, oxygen gas and the noble gases are all part of inorganic chemistry.
3. Analytical Chemistry
Analytical chemistry is concerned with the composition of substances. For instance, an analytical chemist might analyze a sample of blood to see if there is poison present and determine the type of poison.
- Qualitative - Identifies all the atoms and molecules present.
- Quantitative - Amount of each component is determined.
4. Physical Chemistry
Physical chemistry deals with the theoretical basis for the behavior of chemical substances cause of the change that occur. It provides the basis or background for the other types of Chemistry.

5. Biochemistry
Biochemistry studies the processes and compounds important to life. It is closely related to organic chemistry. It studies metabolic processes and reactions like digestion and blood clotting, and biochemical compounds such as carbohydrates, proteins, lipids, nucleic acids.
MADE BY
Michaela Panaguiton and Nadine Leano.




No comments:
Post a Comment